1. The Water Molecule: molecule of water that has two
hydrogen atoms bonded to a single oxygen atom.
2. Solution: a homogeneous mixture of two or more substances.
3. Aqueous Solution: a solution where the solvent is the water
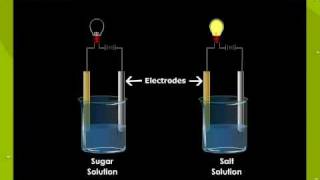
4. Electrolytes: ionic compound that conduct electric current
in aqueous solution, conduct electricity.
5. Non Electrolytes: molecular compounds that do not conduct
electricity.
6. Soluble: a substance that is able to be dissolved in water.
7. Insoluble: a substance that is unable to be dissolved in water.
8. Solute: substances that dissolves into liquid
9. Solvent: can dissolve other substances
hydrogen atoms bonded to a single oxygen atom.
2. Solution: a homogeneous mixture of two or more substances.
3. Aqueous Solution: a solution where the solvent is the water
4. Electrolytes: ionic compound that conduct electric current
in aqueous solution, conduct electricity.
5. Non Electrolytes: molecular compounds that do not conduct
electricity.
6. Soluble: a substance that is able to be dissolved in water.
7. Insoluble: a substance that is unable to be dissolved in water.
8. Solute: substances that dissolves into liquid
9. Solvent: can dissolve other substances
"Strong, Weak, and Non-Electrolytes - Mashpedia Video." Strong, Weak, and Non-Electrolytes - Mashpedia Video. N.p., n.d. Web. 29 May 2014.